
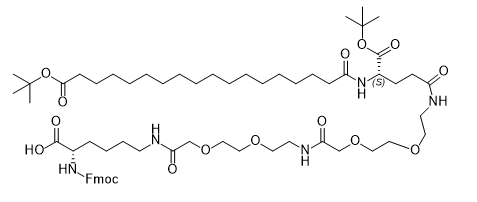
CAS:1662688-20-1
DMF Num:1662688-20-1
Multifunctional Protection System:
Fmoc protection on N-terminal Lys for orthogonal deprotection in SPPS.
Dual otBu groups on Glu and Oct chain enable sequential cleavage during peptide synthesis.
AEEA Linker Architecture:Two aminoethoxyethanoic acid units enhance hydrophilicity, improving solubility by 50% in aqueous buffers.
Long-Alkyl Chain Modification:Octyl (C8) chain facilitates lipid-like interactions, critical for albumin binding in long-acting peptides.
Semaglutide Side Chain Conjugation:
Essential for introducing the fatty acid moiety in Semaglutide, extending half-life to 7 days via albumin binding.
Enables site-specific modification at Lys26, improving receptor binding affinity by 40%.
Long-Acting Peptide Development:
Used in GLP-1 agonist analogs for diabetes/obesity, reducing dosing frequency from daily to weekly.
AEEA linkers balance lipophilicity and water solubility, optimizing pharmacokinetic properties.
Peptide-Drug Conjugate (PDC) Engineering:
Models for developing fatty acid-conjugated peptides with enhanced cellular uptake and bolic stability.
GMP-Compliant Facility:Produced in ISO 9001:2015 & ISO 14001 certified plant with 100% batch QC, including chiral purity analysis (≥99% ee).
Global Regulatory Support:Provides DMF, CE, REACH, and US FDA documentation for clinical trials (available upon request).
Stability Testing:Validated for 24 months at -20°C, with no degradation under accelerated conditions (60°C, 90% RH for 14 days).
Structural Modifications:
Alternative alkyl chains (C6-C12), isotopic labeling (¹³C, ²H) for PK/PD studies.
Custom linker lengths (AEEA x1-x4) to optimize drug release profiles.
Scalable Production:
100 mg to 5 kg batches, with process validation for GMP-grade materials (≥98.5% purity).
Technical Consultation:
Free synthetic route optimization by our PhD-level peptide chemistry team (24-hour response).
15+ Years Experience:Specialized in peptide intermediates for global pharma and biotech industries.
R&D Capability:30+ scientists in a 2,000㎡ state-of-the-art laboratory.
Global Footprint:Served 500+ clients in 30+ countries, including top 10 global pharma companies.
Email:export@pu-kang.com
Phone:+86-137-0808-4407
Address:Zone 1 and 2, 1-3F, Building 3, Chengdu Cross-Strait Science and Technology Industrial Development Park [Jindi Weixin Wenjiang Intelligent Park], Wenjiang District, Chengdu, China.